
Industrial Cleaning Robots for Pharmaceutical Manufacturers in India
In pharmaceutical manufacturing, cleaning is not a routine activity. It is a regulated, quality-critical process. Every floor, corridor, and support area contributes to contamination control. During FDA audits, inspectors do not just examine products—they evaluate systems, discipline, consistency, and documentation. Many pharma companies face audit observations not because they do not clean, but because they cannot prove they cleaned consistently and correctly. This is why industrial cleaning robots for pharma manufacturers in India are becoming essential. They are no longer just about convenience. They are about compliance, traceability, and audit survival.
Table of Contents
- Why GMP Compliance and FDA Audits Are Critical for Pharma Manufacturers?
- How FDA Auditors Evaluate Cleaning in Pharma Facilities?
- Why Manual and Human-Based Cleaning Costs Pharma Manufacturers More Than You Realize?
- Industrial Cleaning Robots for Pharma Manufacturers- The Smarter, Safer and Simpler GMP Strategy
- Why Digital Proof Is Critical for GMP Compliance and FDA Audits?
- Pudu CC1 Pro Is the Best Bet for GMP-Controlled Environments
- How Does CC1 Pro Fit Perfectly for a Pharmaceutical Manufacturing Facility?
- CC1 Pro: Unlimited by Space, Unmatched in Cleanliness
- Pudu Robotics and it’s Revolution with World Class Industrial Cleaning Robots for Pharmaceutical Manufacturers
- Conclusion
Why GMP Compliance and FDA Audits Are Critical for Pharma Manufacturers?

Good Manufacturing Practices (GMP) are extremely mandatory for pharmaceutical manufacturers. They form the foundation of pharmaceutical operations. Any failure in GMP can lead to serious consequences, including warning letters, production halts, product recalls, and long-term brand damage.
Cleaning is deeply connected to GMP because it directly impacts contamination control. Dust, debris, residues, and microbial growth can compromise product safety.
FDA audits focus heavily on how these risks are managed.
Inspectors do not accept verbal explanations. They demand objective evidence.
They want to see:
- Standardized procedures
- Controlled execution
- Verified completion
- Traceable records
If these are missing, the system is considered weak.
How FDA Auditors Evaluate Cleaning in Pharma Facilities?

FDA auditors do not treat cleaning as a housekeeping activity. They treat it as a controlled GMP process—just like manufacturing, packaging, or quality testing.
Their focus is not on how clean the floor looks. Their focus is on process control, consistency, and documentation.
During an audit, they typically look for:
- Clearly defined and approved SOPs
- Area-wise cleaning frequencies (what is cleaned, how often, and why)
- Standardized cleaning methods
- Clear accountability (who is responsible)
- Verifiable proof that cleaning actually happened
They also ask very specific, evidence-based questions such as:
- Who cleaned this area yesterday?
- At what exact time?
- How do you know it was fully cleaned?
- What happens if a scheduled cleaning is missed?
- Where is all this recorded and stored?
If these answers rely on memory, individual staff, or manual paper logbooks, the system is considered high risk.
Why? Because GMP is not built on assumptions—it is built on evidence.
This is where pharmaceutical cleaning automation becomes extremely valuable. Automation removes human dependency, standardizes execution, and creates automatic, time-stamped, audit-ready proof.
And that is exactly what regulators want to see.
Why Manual and Human-Based Cleaning Costs Pharmaceutical Manufacturers More Than You Realize?
In pharmaceutical manufacturing, cleaning is not just an operational activity—it is a compliance-critical process. Yet most facilities still rely heavily on manual, human-based cleaning. This creates hidden costs that go far beyond labor.
Every time a person enters a controlled or cleanroom-adjacent area, the process becomes slow, resource-heavy, and risky. Staff must gown up, wear PPE, follow entry protocols, disinfect, log movements, and then repeat the same steps while exiting. This alone consumes a significant amount of time and manpower.
But the bigger issue is variability. As humans get tired, become hasty during busy shifts, forget essential steps and take shortcuts.
This leads to:
- Inconsistent cleaning quality
- Missed corners and low-visibility zones
- Rework and repeat cleaning
- Increased contamination risk
- Higher chances of audit observations
- Production delays
- Potential batch rejections
From a GMP perspective, this is dangerous.
Manual systems depend on individuals, not controls. And auditors do not evaluate intent—they evaluate process control. If your cleaning depends on people remembering what to do, it is considered a weak system.
This is how cleaning becomes a silent contributor to:
• Audit findings
• CAPAs
• Downtime
• Quality incidents
• Reputation damage
Industrial Cleaning Robots for Pharmaceutical Manufacturers- The Smarter, Safer and Simpler GMP Strategy

Modern pharma manufacturing is moving toward automation not for convenience, but for compliance. Industrial cleaning robots are not just machines—they are process control tools.
Unlike humans, industrial cleaning robots operate with complete consistency, no matter how long the shift or how heavy the workload. They are not affected by fatigue, pressure, or distractions. This makes industrial cleaning robots far more reliable for GMP-controlled environments where predictability and discipline are essential.
- They do not get tired over long shifts.
- They do not rush when workloads increase.
- They do not forget steps in a process.
- They do not skip areas to save time.
- They do not deviate from defined protocols.
Robotic cleaning also reduces human presence in controlled areas, which directly reduces contamination risk. Fewer people entering clean zones means fewer particles, fewer microbes, and fewer variables.

For pharma manufacturers, this means:
- Lower manpower dependency
- Reduced PPE usage
- Less re-cleaning
- Fewer quality deviations
- Faster inspections
- Stronger audit confidence
This is why industrial cleaning robots are no longer optional. They are becoming the only scalable way to maintain GMP discipline without increasing cost, risk, or operational burden.
Why Digital Proof Is Critical for GMP Compliance and FDA Audits?

In pharmaceutical manufacturing, cleaning is not judged by how clean a floor looks. It is judged by how well the process is controlled, documented, and proven.
FDA and GMP systems are evidence-driven. Inspectors do not rely on verbal explanations or assurances. They rely on records, timestamps, logs, and traceable proof. If an activity is not documented, it is treated as if it never happened.
This is where many pharma manufacturers struggle. Manual cleaning systems rely on paper logbooks, memory, or human reporting. These records are often incomplete, difficult to verify, or filled retrospectively. During audits, this creates uncertainty—and uncertainty is considered risk.
Auditors typically look for:
- Time-stamped records
- Area-wise cleaning history
- Proof of coverage
- Method of cleaning used
- Confirmation that the task was completed
- Evidence of consistency over time
When this information cannot be produced instantly and clearly, the system is considered weak.
This is where the Pudu CC1 Pro fundamentally changes the model.
Instead of treating cleaning as a manual activity, it treats it as a digitally controlled process. Every cleaning cycle is automatically logged. Every run is time-stamped. Every route is recorded. Coverage is mapped. Completion is verified. Reports can be exported as PDFs.
This transforms cleaning from an operational task into a documented GMP process.
Industrial Cleaning Robots for Pharmaceutical Manufacturers don’t just possess convenience features. They are compliance enablers.
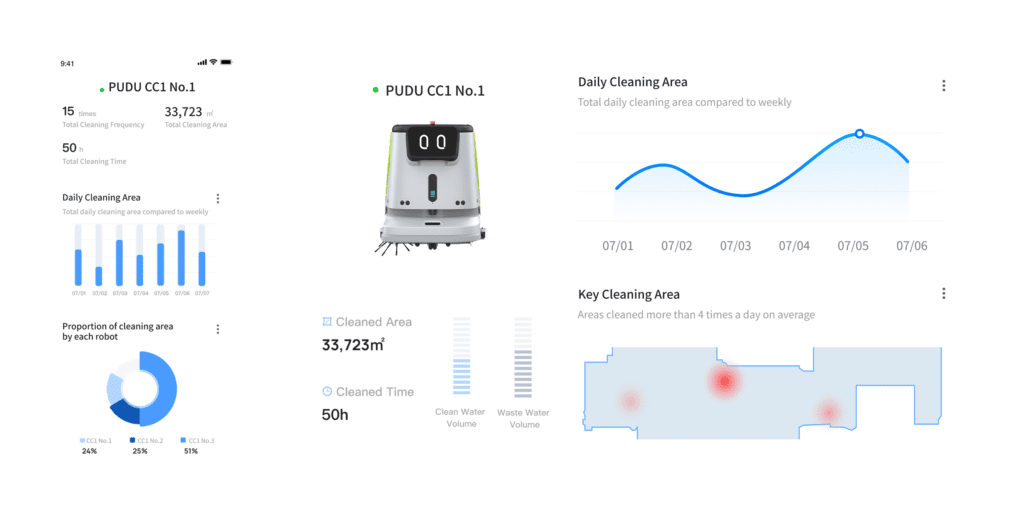
Pudu Link App’s detailed cleaning analysis report
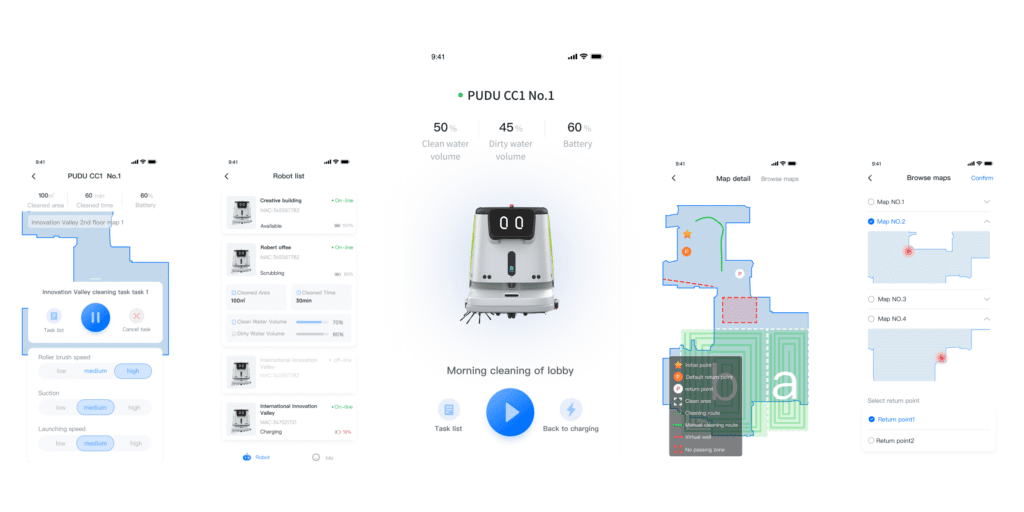
Pudu Link App showing details of tracked maps for cleaning
Pudu CC1 Pro Is the Best Bet for GMP-Controlled Environments

The Pudu CC1 Pro is not just another cleaning robot. It is designed to function as a process control system for regulated environments like pharmaceutical plants. What makes CC1 Pro different is not just that it cleans—but how it cleans.
- It follows fixed, programmed routes.
- It maintains the same speed and coverage every time.
- It does not rush, skip, or improvise.
- It documents every run automatically.
This removes human variability completely.
Instead of depending on memory and paper logbooks, CC1 Pro creates time-stamped, traceable, digital proof of every cleaning task. It records when an area was cleaned, how it was cleaned, how long it took, and whether it was completed.
This is exactly what GMP systems demand.
And this is where Pudu Robotics stands apart.
Pudu is not just selling machines. It is building intelligent, compliance-ready cleaning systems that fit into GMP workflows. Features like AI-powered navigation, heatmaps, route tracking, automated reports, and task history make CC1 Pro far more than a cleaning device—it becomes part of your quality system.
For pharma manufacturers, this means:
- Less dependency on manual labor
- Less contamination risk
- Stronger audit readiness
- Automatic documentation
- Higher confidence during inspections
This is why CC1 Pro is not just a convenience upgrade. It is a compliance upgrade.
And that is why Pudu Robotics is the safest long-term choice for GMP-controlled environments.
How Does CC1 Pro Fit Perfectly for a Pharmaceutical Manufacturing Facility?
Pharmaceutical plants are not uniform spaces. They consist of multiple zones, each with different contamination risks and different GMP expectations.
CC1 Pro is ideal for non-product-contact floor areas that still fall under strict GMP control, including:
- Warehouse aisles and material movement paths
- Pre-production corridors and buffer zones
- Change rooms and gowning areas
- Staging zones
- Production support corridors
- QA offices and administrative walkways
These areas are critical because they act as contamination transfer points. Movement through them spreads particles, debris, and microbes.

CC1 Pro performs:
- Sweeping
- Scrubbing
- Vacuuming
- Mopping
All in one platform.
This eliminates the need for multiple machines, multiple SOPs, and multiple documentation systems. It also allows cleaning practices to be standardized across the entire facility.
CC1 Pro: Unlimited by Space, Unmatched in Cleanliness
In GMP environments, “looking clean” is not enough. What matters is whether the process is controlled, repeatable, and verifiable. This is only possible with Industrial Cleaning Robots for Pharma Manufacturers in India.
CC1 Pro uses a combination of Visual SLAM and Laser SLAM navigation. This allows it to map areas precisely, follow the same routes every time, and avoid shortcuts.
 This ensures:
This ensures:
- Fixed cleaning paths
- Predictable movement
- Consistent coverage
- No skipped zones
It also includes performance features designed for industrial-grade cleaning:
- Stronger suction for fine particles
- Faster brush rotation for deeper scrubbing
- Spot scrubbing for stubborn residues
- AI-based detection of heavily soiled areas
- Automatic re-cleaning of missed or dirty zones
This is not cosmetic cleaning. It is controlled, repeatable, and verifiable cleaning. CC1 Pro does not just clean. It verifies.
CC1 Pro Delivers Audit-Ready Digital Proof for GMP Compliance
For pharmaceutical plants using industry cleaning robots for pharmaceutical manufacturers, proof matters as much as performance. CC1 Pro generates automatic, detailed cleaning reports that QA teams use as direct GMP evidence during FDA audits.
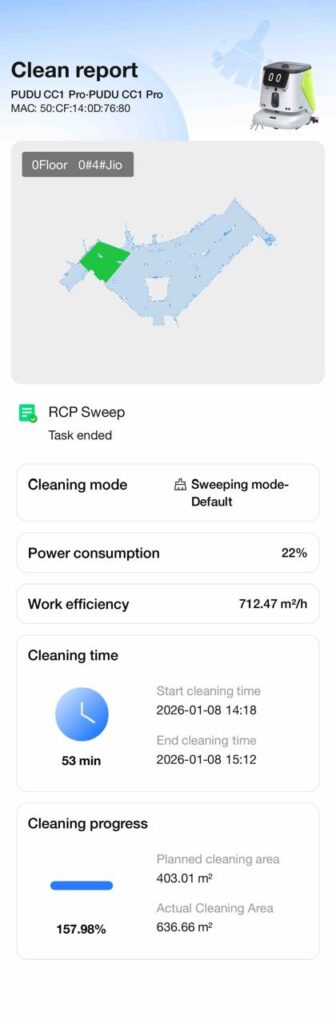
Each report records:
- Exact cleaning routes and coverage maps
- Start and end time of every task
- Cleaning mode used (sweeping, silent dust pushing, etc.)
- Planned vs. actual area cleaned
- Work efficiency (m²/hour)
- Task completion status
- Power consumption
Instead of relying on manual logbooks or staff memory, pharma teams present these reports as objective, tamper-resistant proof. Auditors can clearly see what was cleaned, when it was cleaned, how it was cleaned, and how thoroughly it was done.
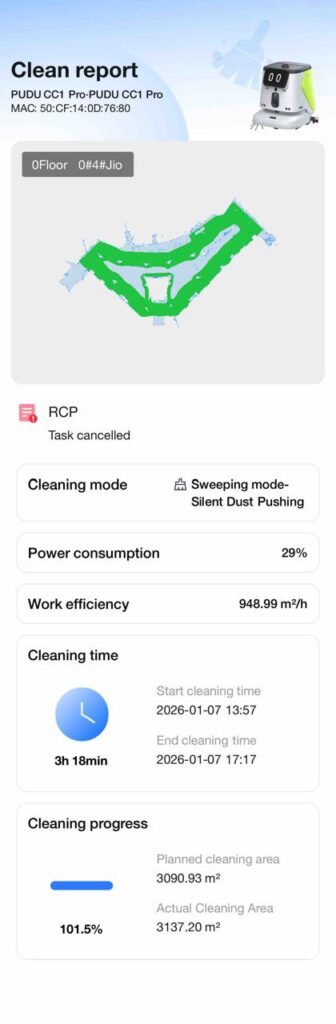
This turns cleaning into a controlled, verifiable GMP process—not just a housekeeping task.
Pudu Robotics and it’s Revolution with World Class Industrial Cleaning Robots for Pharmaceutical Manufacturers
Pudu CC1 – For General and Non-Critical Areas


The Pudu CC1 is a versatile 4-in-1 cleaning robot designed for daily maintenance in non-critical or semi-controlled zones.
It supports:
- Sweeping
- Scrubbing
- Vacuuming
- Mopping
Key strengths:
- Multi-mode cleaning in a single machine
- Manual ergonomic handle for quick interventions
- Visual + Laser SLAM navigation
- Digital task tracking
- Automatic charging and water refill/drain (with docking)
- Breakpoint resume cleaning if battery runs low
This makes CC1 suitable for:
- Admin corridors
- Utility zones
- Secondary walkways
- Non-critical storage areas
It reduces manual labor while maintaining baseline hygiene—but it is not designed for strict GMP documentation workflows like CC1 Pro.
Pudu MT1 Series – For Ultra-Large Warehouses and Logistics Zones


The MT1 series is built for scale.
These robots are designed to handle massive floor areas with heavy dust loads, pallet movement, and continuous operation requirements.
Key strengths:
- High-capacity waste collection
- AI + VSLAM navigation
- Strong sweeping performance
- Long runtimes
- Designed for 24/7 operation
- Built for industrial durability
MT1 variants can include:
- Vacuuming options
- HEPA filtration (in select models)
- Night operation
- High obstacle tolerance
Ideal use cases:
- Raw material warehouses
- Finished goods warehouses
- Dispatch zones
- High-traffic logistics floors
These zones often generate dust from cartons, pallets, and forklifts. MT1 robots keep these areas under control so contamination does not migrate into production.
Pudu SH1 – For Narrow Corridors and Compact Spaces


SH1 is a compact, upright scrubber-style robot designed for tight spaces.
Key strengths:
- Slim profile for narrow corridors
- Strong scrubbing pressure
- High brush rotation speed
- Efficient water usage
- High suction power
- Easy maneuverability
It is suitable for:
- Tight corridors
- Change rooms
- Small rooms
- Utility spaces
- Transitional zones
SH1 helps automate areas where large robots cannot move easily, reducing manual cleaning dependency.
Conclusion
In today’s regulatory environment, pharmaceutical manufacturers need more than manual processes—they need controlled, traceable, and audit-ready systems. Pudu Robotics, together with Autofina Robotics as its official Indian distribution partner, hence delivers the best in class Industrial Cleaning Robots for Pharmaceutical Manufacturers.
With CC1 Pro and its digital proof capabilities, pharma manufacturers turn cleaning into a GMP-aligned, verifiable process instead of a compliance risk. This partnership actively reduces audit stress, contamination risk, and compliance uncertainty—making GMP readiness simpler, stronger, and more reliable.
